HOW TO PICK THE RIGHT DRUG REGULATORY AFFAIRS PROFESSIONAL
“Know thy self” is as important in life as it is in the development lifecycle of a health product – be it a drug (pharmaceutical, biologic or biotechnical), a medical device or other; particularly as you embark on finding the “perfect” fit in the regulatory employee or consultant you bring on.
In fact, the accelerated use of technologies in life sciences, such as stem cell research, gene therapy, real world evidence, mobile medical devices, and cloud computing to name a few, underscores the necessity for picking the right regulatory affairs professional.
MEETING THE CHALLENGES AHEAD
Organizations in the Life Sciences continue to advance effective risk and compliance programs, for instance, by incorporating cyber security practices to safeguard patient data as well as corporate assets. As this new landscape emerges, regulatory affairs professionals will, more than ever, need to be agile and informed enough to meet upcoming and unexpected challenges.
Determining who that regulatory professional is depends largely on your goals. Regulatory professionals must be skilled at taking into consideration complex global dimensions – including multinational supply chains for product manufacturing, global shipping, sales, and various marketing and post-approval surveillance requirements for multiple regions.
In January 2016, the FDA released post-market guidance outlining its cyber security expectations for medical device manufactures already in the field and in the pipeline. Other similar initiatives are being either considered or implemented in various countries.
As of January 2019, agencies such as Health Canada require companies to undergo MDSAP audits. knowledge in this area is essential as companies react to the slow adoption of a global regulatory assessment process.
Because the health product/medical device development process is a complex and ever-changing one; you want to be confident and at ease with who you will have chosen to be at the helm. Here, we will look at what credentials matter, where to find Regulatory Professionals, and fit (i.e. how well the person is likely to interface with your team).
WHAT CREDENTIALS MATTER
Most people who make the transition into the regulatory profession have relevant prior experience in related fields (e.g., research and development, quality or a clinical profession). Typically, they will hold at minimum a bachelor’s degree in science, preferably in chemistry, biochemistry, pharmacy, pharmacology, or pharmaceutical technology. However, this alone doesn’t make a good regulatory affairs professional.
There are several organizations and university departments that offer training / certifications in regulatory affairs, whether the candidate is experienced or not. Look for a professional who has the following credentials or training when hiring:
-Regulatory Affairs Certification, the RAC credential, is a well reputed, accredited, post-academic, professional credential for the regulatory professional working in the healthcare product arena, overseen by RAPS.
-RAPS (Regulatory Affairs Professionals Society) is a renowned organization that offer certification in regulatory affairs for medical devices and/or pharmaceuticals. Their numerous online courses cover essential concepts of healthcare product regulation and regulatory issues at each stage of the product lifecycle and for different regions of the world.
–D.É.S.S. en développement du médicament of the University of Montreal, offers courses specific to the global health product development.
-AAPS (Academy of Applied Pharmaceutical Sciences) This certification program provides the comprehensive background and training required of regulatory affairs professionals to address domestic and international regulatory statutes and laws. Its program includes courses on International, Health Canada, and FDA’s laws, regulations, and Guidelines, investigational and marketing applications, technical writing, negotiation skills, development of New Drug Application (NDA) submissions, labeling and drug Information, Common Technical Documents (CTD), Notice of Compliance (NOC), Good Clinical Practices (GCPs), requirements for ongoing post-marketing surveillance and post-marketing changes, communication and management skills essential for the successful regulatory affairs professional in an industry work environment.
Now, as helpful as a candidate’s background or certifications may be, there is one thing that trumps both: experience.
In an industry that is as ever-changing and complex as this one, professionals with a vast amount of experience bring to the table the most valuable skills, knowledge, insight and results.
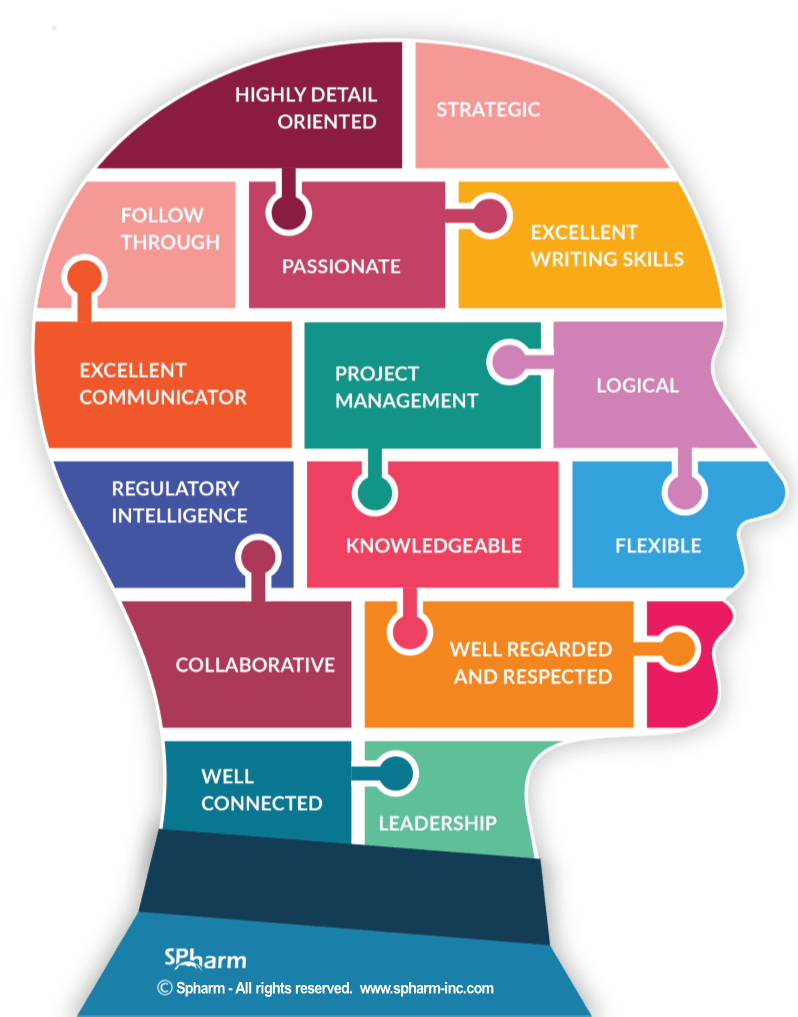
WHAT TRAITS MATTER MOST
There are attributes that are essential parts of all roles and positions. In regulatory affairs, the traits of a candidate matter as much as their experience.
We delved into the role of a regulatory affairs expert and outlined the traits necessary for an excellent professional. You can read the full article here. In the meantime, these are some of the skills and traits to look for:
TRAITS: Collaborative Leader, Logical, Analytical, Meticulous, Collaborative & Flexible, Diligent, Passionate
SKILLS: Able to perform under stress, Regulatory Intelligence, Project Management, Excellent verbal & written communication
WHERE TO FIND QUALIFIED REGULATORY PROFESSIONALS
-RAPs, AAPS, may be able to direct you to a list of graduates.
-LinkedIn is an excellent platform to find professionals who have the experience and certification you are looking for, in particular, look at the various groups within the industry such as:
- CAPRA – Canadian Association of Professionals in Regulatory Affairs
- Clinical Research Canada
- Clinical Research Professionals
- Drug Regulatory Affairs
- Pharma Connection Worldwide
- Professionals in the Pharmaceutical and Biotech Industry
- RADSP – Regulatory Affairs & Drug Safety Professionals
- Total Orphan Drug
-When hiring an individual to join your staff, look at the scope and length of experience directly associated to the work you need done. This is a role that a person needs to come in knowing how to do well, not a role that they can take time learning on the job. You’ll want someone seasoned and well connected; someone with the ability to adapt to such difficult circumstances. Flexibility too, is a desirable quality for working in such a complex and changeable landscape; it also happens to be one of the cornerstones of successful consultancies.
-When looking to hire an independent consultant or an outside firm like SPharm, understand that these consultants have gained a range of experiences in different markets and industries, allowing them to bring diversity of thought, experience and expertise to clients. They have also established a strong network relationships within the industry through dealing with multiple clients, colleagues and regulatory agencies. This network is an invaluable asset.
-When hiring a regulatory consultancy, much like SPharm, look at the profiles of their staff and how the team as a whole reflects the necessary broadness of skills and experience. Read any articles, posts and reports they produce because they will demonstrate depth of knowledge. If there are webinars, slide decks, whitepapers, eguides or books they have prepared or authored, watch those to also gauge their depth.
By its very nature, a regulatory consultancy must be absolutely well versed and informed on any and all regulatory specific changes, both locally and internationally. This knowledge presents an enormous advantage and opportunity for businesses seeking to hire them.
In addition to the traits above and the requisite skills and experience, you want to know that the person or company you hire has the respect and recognition of Regulatory Agencies and peers. A large and important aspect of the job is facilitating and navigating the complex regulatory landscape.
FIT: A MATCH MADE IN REGULATORY HEAVEN
Finding the right regulatory professional has less to do with the role itself and more to do with alignment to your company culture. This is where the notion of “Know Thy Self” becomes particularly important.
When hiring an individual, look at whether their aspirations match the job. Does the candidate fit into your growth plan for your business beyond filling a need you have right now? How do they want to grow their career in next several years and do they think this job with you can help them fulfill their aspirations? Getting a sense of both aspects will help you determine whether or not there is alignment between the candidate and your company’s goals.
Next, vet them appropriately. Applicable to both candidates and consultancies, it is important to understand how they work and who they are as a co-worker/service provider. You know the experience and traits you’re looking for, now it’s a matter of matching their past performance to the desired output you want. This involves vetting their references and asking pertinent questions to get an idea of their capabilities and work ethic.
Pay attention to the questions they ask; their questions show preparedness and engagement on the part of the candidate. The best hires care about the team they’ll be on or the company they are providing services to; they care about how they can help take your company forward. Questions will demonstrate their knowledge of the industry, and how apt they are – especially service providers – to understand the unique needs and goals of your organization.
Finally, be crystal clear about expectations. They might end up wearing several hats and going above and beyond. Working in the regulatory space isn’t easy, and while it is rewarding, it’s important to make sure candidates know they are going to work in frequent shifting conditions.
Always trust your instincts. You might be in a hurry to hire but hiring right matters more than hiring now.

With a quarter of a century of consistent and successful results, SPharm has become one of the leading Drug Regulatory Advisory firms in Canada. Drug companies from around the world count on SPharm to help them navigate the drug (and other health products) review & approval process in Canada.
Avec un quart de siècle de résultats et de succès constants , SPharm est devenue une entreprise de services de référence au Canada en stratégie de développement de médicaments et affaires réglementaires. Des compagnies pharmaceutiques à travers le monde comptent sur SPharm pour faciliter le processus d’homologation de leurs médicaments (et autres produits de santé) au Canada.